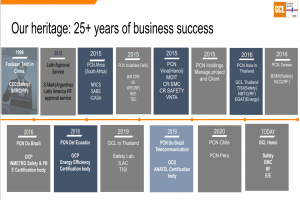
ANVISA certification in Brasil
PCN do Brasil is the branch of our company in the Latin American market, recognized by the agency Cgcre-Inmetro/IAF as a Certification Body according to the product range of the accreditation number nº OCP 0104:

Home Appliances - Safety, Energy and EMC
Inmetro Ordinance 371/2009
IEC 60335-1 Series
Lighting Equipment – Safety, EMC and Energy Efficiency
Inmetro Ordinance RTQ 144/2012
IEC 62031/2013
IEC 62560/2013
IEC 62612/2013
CISPR 15
Medical devices
IEC 60601 Series + Syringes / Needles / Infusion Sets
Telecommunications equipment
Categories I, II and III
Inmetro Ordinance 371/2009
IEC 60335-1 Series
Lighting Equipment – Safety, EMC and Energy Efficiency
Inmetro Ordinance RTQ 144/2012
IEC 62031/2013
IEC 62560/2013
IEC 62612/2013
CISPR 15
Medical devices
IEC 60601 Series + Syringes / Needles / Infusion Sets
Telecommunications equipment
Categories I, II and III
ANVISA Certification
Agencia Naciona Vigilancia Sanitaria : Cơ quan quản lý y tế Brazil (ANVISA)
Depending on the item, Brazilian medical device certification is divided into compulsory and non-mandatory certification. Brazil's medical device classification system is similar to Europe's fundamental regulation 93/42/EEC. ANVISA requires medical device manufacturers to comply with Brazilian Good Manufacturing Practices (GMP), and INMETRO certification is further required for electrically powered devices.
The body authorized to issue product certification, called Product Certification Bodies (Organismos de Certificação de Produto - OCP), must be accredited by the CGCRE, based on the principles and policies adopted within the SBAC.
The choice of the test laboratory follows the rule defined in the Conformity Assessment Regulations, but prioritizes the execution of tests in laboratories Accredited by the CGCRE.
In order to import or sell medical devices in Brazil, the manufacturer or importer must go through the medical device registration process with ANVISA and must have a BRH (Brazilian Registration Holder). Depending on the risk of the product, it is divided into 4 different classes.
Target Product:
All medical devices are subject to registration and are classified as Class I, Class II, Class III or Class IV (Brazilian Resolution RDC 185/2001)
- Class I: Products that are unlikely to pose a risk to users or public health and require only ANVISA summary registration
- Class 2 (Classe II): Products that are likely to pose a risk to users or public health and require official registration with ANVISA
- Class 3 (Classe III): Products with a very high possibility of providing risks to users or public health require official registration with ANVISA
- Group 3-a (Classe IV): Items that require auto test and require official registration with ANVISA
Agencia Naciona Vigilancia Sanitaria : Cơ quan quản lý y tế Brazil (ANVISA)
Depending on the item, Brazilian medical device certification is divided into compulsory and non-mandatory certification. Brazil's medical device classification system is similar to Europe's fundamental regulation 93/42/EEC. ANVISA requires medical device manufacturers to comply with Brazilian Good Manufacturing Practices (GMP), and INMETRO certification is further required for electrically powered devices.
The body authorized to issue product certification, called Product Certification Bodies (Organismos de Certificação de Produto - OCP), must be accredited by the CGCRE, based on the principles and policies adopted within the SBAC.
The choice of the test laboratory follows the rule defined in the Conformity Assessment Regulations, but prioritizes the execution of tests in laboratories Accredited by the CGCRE.
In order to import or sell medical devices in Brazil, the manufacturer or importer must go through the medical device registration process with ANVISA and must have a BRH (Brazilian Registration Holder). Depending on the risk of the product, it is divided into 4 different classes.
Target Product:
All medical devices are subject to registration and are classified as Class I, Class II, Class III or Class IV (Brazilian Resolution RDC 185/2001)
- Class I: Products that are unlikely to pose a risk to users or public health and require only ANVISA summary registration
- Class 2 (Classe II): Products that are likely to pose a risk to users or public health and require official registration with ANVISA
- Class 3 (Classe III): Products with a very high possibility of providing risks to users or public health require official registration with ANVISA
- Group 3-a (Classe IV): Items that require auto test and require official registration with ANVISA
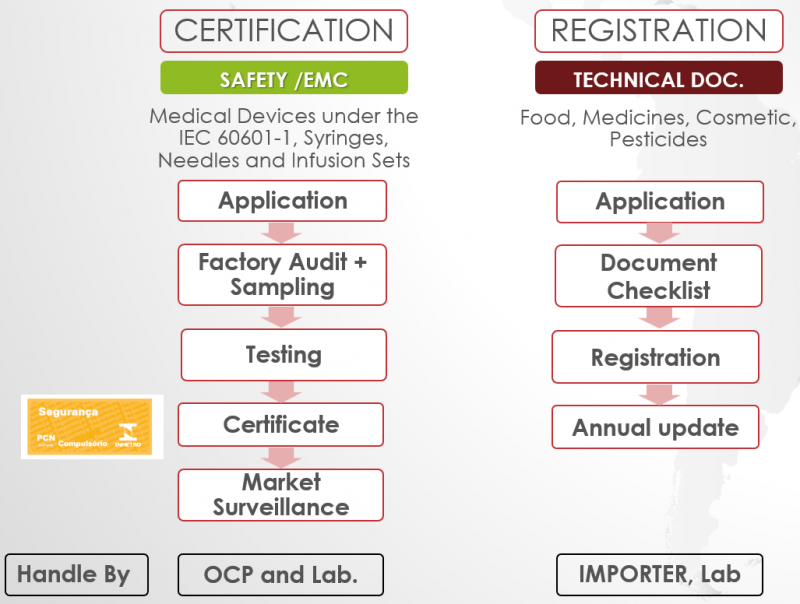
Below is the general certification process for products under the ANVISA Certification and Registration category
Below is the ANVISA Certificate

Contact information for consultation:
GCL (HCM) COMPANY LIMITED
Hotline: 0393423091 | Hanoi: 02473099929
Email: hcm@gclab.org | louise.vietnam@pcn-global.com
Office: No. 27, Street 12, Khang Dien Residential Area, Phuoc Long B Ward, Thu Duc City, Ho Chi Minh City
Testing Lab: No. 115, An Phu Alley 04, An Phu Ward, Thuan An City, Binh Duong Province.
GCL (HCM) COMPANY LIMITED
Hotline: 0393423091 | Hanoi: 02473099929
Email: hcm@gclab.org | louise.vietnam@pcn-global.com
Office: No. 27, Street 12, Khang Dien Residential Area, Phuoc Long B Ward, Thu Duc City, Ho Chi Minh City
Testing Lab: No. 115, An Phu Alley 04, An Phu Ward, Thuan An City, Binh Duong Province.


.png)